When a comet approaches the Sun, it receives more heat so that more cometary ices and dust are released. The major neutral gas released by the comet is H2O but there are plenty of other fish in the sea!
Hiding hydrogen halides
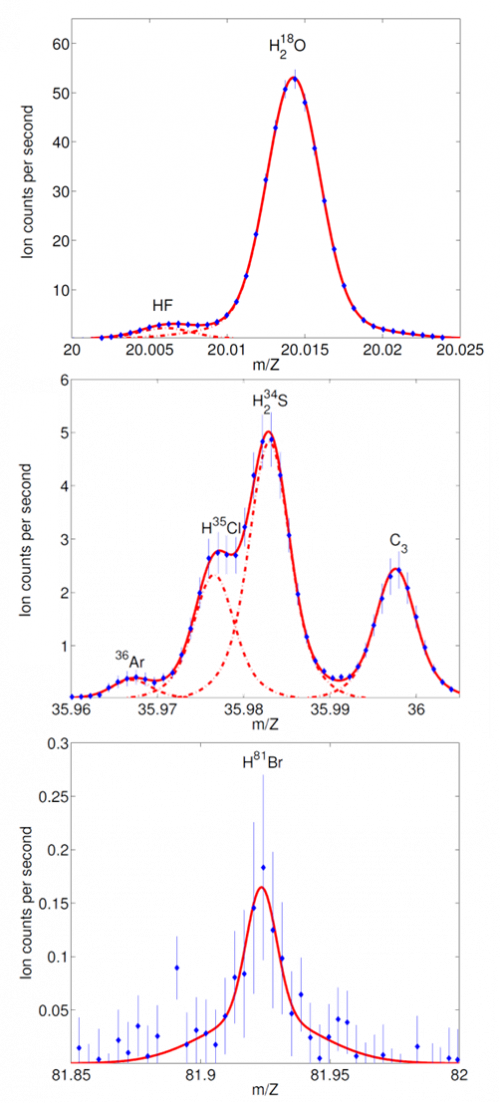
Halogens like fluorine (F), chlorine (Cl) and bromine (Br) exist in several forms on Earth. The halogen-containing compound we are most familiar with is sodium chloride (NaCl) or kitchen salt. Using the mass spectrometer DFMS on Rosetta we could investigate whether or not halogens are present in the atmosphere surrounding comet 67P. Halogens are mainly found in the form of hydrogen halides. Some are hiding next to other species in the spectra (like HF and HCl) and some are very lowly abundant (like HBr), as illustrated in Figure 2. This made discovering them quite a challenge!
Not uniformly frozen in the water ice on the comet
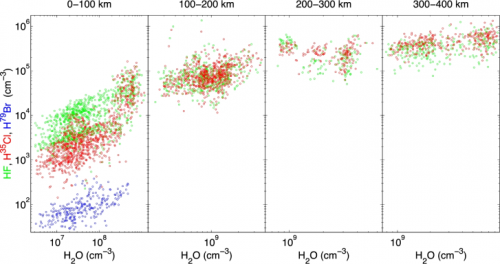
Remarkably, the amounts of hydrogen halides released from the comet vary with the amount of water, but are not linearly proportional to it. This proves that these species are not uniformly frozen in the water ice on the comet. So where do the hydrogen halides then come from? Two important observations hint to the answer.
- The hydrogen halide contribution relative to water increases as a function of the distance from the comet in the first few 100 kilometers.
- The farther away, the less the hydrogen halide abundance seems to depend on variations in the amount of water, as illustrated in Figure 3.
The only plausible explanation is that the hydrogen halides reside in the inner ice mantles around cometary dust grains, while the outer mantles consist of rather pure water ice; comet dust particles are known to consist of aggregates of such grains, as in Figure 4. The ice that sublimates while the dust is still on the comet surface is mostly the pure water ice in the outer mantles, so that there are not many halides close to the comet. As the dust particles are released from the comet, they heat up and both their pure ice and halogen-rich mantles evaporate, so that the fraction of halides increases with distance.